Command line
Assuming you built the Nyxus executable from source code, the following parameters are available for the command line usage. Regular command line users should adhere parameter value to the “Type” column. WIPP developers should adhere to columns “WIPP I/O role” and “WIPP type”.
Parameter |
Description |
Type |
WIPP I/O role |
WIPP type |
|---|---|---|---|---|
–outputType |
Output type for feature values. Acceptable value: speratecsv, singlecsv, arrow, parquet. Default value: ‘–outputType=separatecsv’ |
string constant |
input |
enum |
–features |
String constant or comma-seperated list of constants requesting a group of features or particular feature. Default value: ‘–features=*ALL*’ |
string |
input |
array |
–filePattern |
Regular expression to match image files in directories specified by parameters ‘–intDir’ and ‘–segDir’. To match all the files, use ‘–filePattern=.*’ |
string |
input |
string |
–intDir |
Directory of intensity image collection |
path |
input |
collection |
–outDir |
Output directory |
path |
output |
csvCollection |
–segDir |
Directory of labeled image collection |
path |
input |
collection |
–coarseGrayDepth |
(Optional) Custom number of grayscale level bins used in texture features. Default: ‘–coarseGrayDepth=256’ |
integer |
input |
integer |
–gaborfreqs |
(Optional) Feature GABOR: custom denominators of \(\pi\) as frequencies of Gabor filter’s harmonic factor. Default: ‘–gaborfreqs=1,2,4,8,16,32,64’ |
list of integer constants |
input |
collection |
–gaborf0 |
(Optional) Feature GABOR: frequency of the baseline lowpass filter as denominator of \(\pi\). Default: ‘–gaborf0=0.1’ |
real |
input |
number |
–gaborgamma |
(Optional) Feature GABOR: aspect ratio of Gabor filter’s Gaussian factor. Default: ‘–gaborgamma=0.1’ |
real |
input |
number |
–gaborkersize |
(Optional) Feature GABOR: dimension of the 2D Gabor filter kernel. Default: ‘–gaborkersize=16’ |
integer |
input |
integer |
–gaborsig2lam |
(Optional) Feature GABOR: spatial frequency bandwidth of Gabor filter. Default: ‘–gaborsig2lam=0.8’ |
real |
input |
number |
–gabortheta |
(Optional) Feature GABOR: orientation of the Gaussian in degrees 0-180. Default: ‘–gabortheta=45’ |
real |
input |
number |
–gaborthold |
(Optional) Feature GABOR: lower threshold of the filtered image to baseline ratio. Default: ‘–gaborthold=0.025’ |
real |
input |
number |
–glcmAngles |
(Optional) Feature GLCM: enabled direction angles. Superset of values: 0, 45, 90, and 135. Default: ‘–glcmAngles=0,45,90,135’ |
list of integer constants |
input |
collection |
–intSegMapDir |
(Optional) Data collection of the ad-hoc intensity-to-mask file mapping. Must be used in combination with parameter ‘–intSegMapFile’ |
path |
input |
collection |
–intSegMapFile |
(Optional) Name of the text file containing an ad-hoc intensity-to-mask file mapping. The files are assumed to reside in corresponding intensity and label collections. Must be used in combination with parameter ‘–intSegMapDir’ |
string |
input |
string |
–pixelDistance |
(Optional) Number of pixels to treat ROIs within specified distance as neighbors. Default value: ‘–pixelDistance=5’ |
integer |
input |
integer |
–pixelsPerCentimeter |
(Optional) Number of pixels in centimeter used by unit length-related features. Default value: 0 |
real |
input |
number |
–ramLimit |
(Optional) Amount of memory not to exceed by Nyxus, in megabytes. Default value: 50% of available memory. Example: ‘–ramLimit=2000’ to use 2,000 megabytes |
integer |
input |
integer |
–reduceThreads |
(Optional) Number of CPU threads used on the feature calculation step. Default: ‘–reduceThreads=1’ |
integer |
input |
integer |
–skiproi |
(Optional) Skip ROIs having specified labels. Example: ‘–skiproi=image1.tif:2,3,4;image2.tif:45,56’ |
string |
input |
string |
–tempDir |
(Optional) Directory used by temporary out-of-RAM objects. Default value: system temporary directory |
path |
input |
path |
–arrowOutputType |
(Optional) Type of Arrow file to write the feature results to. Current options are ‘arrow’ for Arrow IPC or ‘parquet’ for Parquet |
string |
output |
enum |
Examples
This chapter presents some particular usage cases of Nyxus
1. Requesting specific features
Suppose we need to extract only Zernike features and first 3 Hu’s moments:
./nyxus --features=ZERNIKE2D,HU_M1,HU_M2,HU_M3 --intDir=/home/ec2-user/data-ratbrain/int --segDir=/home/ec2-user/data-ratbrain/seg --outDir=/home/ec2-user/work/OUTPUT-ratbrain --filePattern=.* --outputType=singlecsv
2. Requesting specific feature groups
Suppose we need to extract only intensity features basic morphology features:
./nyxus --features=*all_intensity*,*basic_morphology* --intDir=/home/ec2-user/data-ratbrain/int --segDir=/home/ec2-user/data-ratbrain/seg --outDir=/home/ec2-user/work/OUTPUT-ratbrain --filePattern=.* --outputType=singlecsv
3. Mixing specific feature groups and individual features
Suppose we need to extract intensity features, basic morphology features, and Zernike features:
./nyxus --features=*all_intensity*,*basic_morphology*,zernike2d --intDir=/home/ec2-user/data-ratbrain/int --segDir=/home/ec2-user/data-ratbrain/seg --outDir=/home/ec2-user/work/OUTPUT-ratbrain --filePattern=.* --outputType=singlecsv
4. Specifying a feature list from with a file instead of command line
Sometimes a list of requested features can be long making Nyxus command line huge. An alternative to dealing with a long command line is specifying all the desired features in a comma, space, and newline delimited text file. Suppose a feature set is in file feature_list.txt:
mean,min,kurtosis
skewness
Then the command line will be:
./nyxus --features=feature_list.txt --intDir=/home/ec2-user/data-ratbrain/int --segDir=/home/ec2-user/data-ratbrain/seg --outDir=/home/ec2-user/work/OUTPUT-ratbrain --filePattern=.* --outputType=singlecsv
5. Whole-image feature extraction
The regular operation mode of Nyxus is processing pairs of intensity and mask images treating non-zero pixel values of the mask image as segment label. The other operation mode is the so called “single-ROI mode” - treating the intensity image as segment. To activate it, just reference the intensity image collection as mask in the command line:
./nyxus --features=*basic_morphology* --intDir=/home/ec2-user/data-ratbrain/int --segDir=/home/ec2-user/data-ratbrain/int --outDir=/home/ec2-user/work/OUTPUT-ratbrain --filePattern=.* --outputType=singlecsv
6. Regular and ad-hoc mapping between intensity and mask image files
Intensity and mask image collections are specified in the command line (via parameters –intDir and –segDir) and the default mapping between an intensity and mask image, after applying a file name pattern (via parameter –filePattern), is the 1:1 mapping:
intensity_image_1 segment_image_1
intensity_image_2 segment_image_2
intensity_image_3 segment_image_3
intensity_image_4 segment_image_4
Here, each intensity and mask image is assumed to reside in the corresponding image collection directory specified with command line arguments –intDir=/home/ec2-user/data-ratbrain/int –segDir=/home/ec2-user/data-ratbrain/seg. More precisely:
/home/ec2-user/data-ratbrain/int/image_1.ome.tif /home/ec2-user/data-ratbrain/seg/image_1.ome.tif
/home/ec2-user/data-ratbrain/int/image_2.ome.tif /home/ec2-user/data-ratbrain/seg/image_2.ome.tif
/home/ec2-user/data-ratbrain/int/image_3.ome.tif /home/ec2-user/data-ratbrain/seg/image_3.ome.tif
/home/ec2-user/data-ratbrain/int/image_4.ome.tif /home/ec2-user/data-ratbrain/seg/image_4.ome.tif
In case the dataset is based on a 1:N mapping, for example
intensity_image_1 segment_image_A
intensity_image_2 segment_image_A
intensity_image_3 segment_image_A
intensity_image_4 segment_image_B
the user needs to pass such an ad-hoc mapping to Nyxus via referenceing a mapping definition text file in the command line (parameter –intSegMapFile).
Note: the order of mapping definition file columns is critical, and the 1-st column is interpreted as the intensity image files column while the 2-nd column is interpreted as the mask image files.
Assuming contents of file mapping.txt is
image_1.ome.tif image_A.ome.tif
image_2.ome.tif image_A.ome.tif
image_3.ome.tif image_A.ome.tif
image_4.ome.tif image_B.ome.tif
and the file is passed to Nyxus via parameter –intSegMapFile, the mapping will resolve to mapping
/home/ec2-user/data-ratbrain/int/image_1.ome.tif /home/ec2-user/data-ratbrain/seg/image_A.ome.tif
/home/ec2-user/data-ratbrain/int/image_2.ome.tif /home/ec2-user/data-ratbrain/seg/image_A.ome.tif
/home/ec2-user/data-ratbrain/int/image_3.ome.tif /home/ec2-user/data-ratbrain/seg/image_A.ome.tif
/home/ec2-user/data-ratbrain/int/image_4.ome.tif /home/ec2-user/data-ratbrain/seg/image_B.ome.tif
7. Ad-hoc mapping between intensity and mask image files via Python interface
Alternatively, Nyxus can process explicitly defined pairs of intensity-mask images, for example image “i1” with mask “m1” and image “i2” with mask “m2”:
from nyxus import Nyxus
nyx = Nyxus(["*ALL*"])
features = nyx.featurize_files(
[
"/path/to/images/intensities/i1.ome.tif",
"/path/to/images/intensities/i2.ome.tif"
],
[
"/path/to/images/labels/m1.ome.tif",
"/path/to/images/labels/m2.ome.tif"
])
Nyxus can also process intensity-mask pairs that are stored as Numpy arrays using the featurize method. This method takes in either a single pair of 2D intensity-mask pairs or a pair of 3D arrays containing 2D intensity and mask images. There is also two optional parameters to supply names to the resulting dataframe, .
from nyxus import Nyxus
import numpy as np
nyx = Nyxus(["*ALL*"])
intens = [
[[1, 4, 4, 1, 1],
[1, 4, 6, 1, 1],
[4, 1, 6, 4, 1],
[4, 4, 6, 4, 1]],
[[1, 4, 4, 1, 1],
[1, 1, 6, 1, 1],
[1, 1, 3, 1, 1],
[4, 4, 6, 1, 1]]
]
seg = [
[[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1]],
[[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[0, 1, 1, 1, 1],
[1, 1, 1, 1, 1]]
]
features = nyx.featurize(intens, seg)
The features variable is a Pandas dataframe similar to what is shown below.
mask_image intensity_image label MEAN MEDIAN ... GABOR_6
0 Segmentation1 Intensity1 1 45366.9 46887 ... 0.873016
1 Segmentation1 Intensity1 2 27122.8 27124.5 ... 1.000000
2 Segmentation1 Intensity1 3 34777.4 33659 ... 0.942857
3 Segmentation1 Intensity1 4 35808.2 36924 ... 0.824074
... ... ... ... ... ... ... ...
14 Segmentation2 Intensity2 6 54573.3 54573.3 ... 0.980769
Note that in this case, default names were provided for the mask_image and intensity_image columns. To supply names for these columns, the optional arguments intensity_names and label_names are used by passing lists of names in. The length of the lists must be the same as the length of the mask and intensity arrays. To name the images, use
intens_names = ['custom_intens_name1', 'custom_intens_name2']
seg_names = ['custom_seg_name1', 'custom_seg_name2']
features = nyx.featurize(intens, seg, intens_name, seg_name)
The features variable will now use the custom names, as shown below
mask_image intensity_image label MEAN MEDIAN ... GABOR_6
0 custom_seg_name1 custom_intens_name1 1 45366.9 46887 ... 0.873016
1 custom_seg_name1 custom_intens_name1 2 27122.8 27124.5 ... 1.000000
2 custom_seg_name1 custom_intens_name1 3 34777.4 33659 ... 0.942857
3 custom_seg_name1 custom_intens_name1 4 35808.2 36924 ... 0.824074
... ... ... ... ... ... ... ...
14 custom_seg_name2 Intensity2 6 54573.3 54573.3 ... 0.980769
All parameters to configure Nyxus are available to set within the constructor. These parameters can also be updated after the object is created using the set_params method. This method takes in keyword arguments where the key is a valid parameter in Nyxus and the value is the updated value for the parameter. For example, to update the coarse_gray_depth to 256 and the gabor_f0 parameter to 0.1, the following can be done:
from nyxus import Nyxus
nyx = Nyxus(["*ALL*"])
intensityDir = "/path/to/images/intensities/"
maskDir = "/path/to/images/labels/"
features = nyx.featurize_directory (intensityDir, maskDir)
nyx.set_params(coarse_gray_depth=256, gabor_f0=0.1)
A list of valid parameters is included in the documentation for this method.
To get the values of the parameters in Nyxus, the get_params method is used. If no arguments are passed to this function, then a dictionary mapping all of the variable names to the respective value is returned. For example,
from nyxus import Nyxus
nyx = Nyxus(["*ALL*"])
intensityDir = "/path/to/images/intensities/"
maskDir = "/path/to/images/labels/"
features = nyx.featurize_directory (intensityDir, maskDir)
print(nyx.get_params())
will print the dictionary
{'coarse_gray_depth': 256,
'features': ['*ALL*'],
'gabor_f0': 0.1,
'gabor_freqs': [1.0, 2.0, 4.0, 8.0, 16.0, 32.0, 64.0],
'gabor_gamma': 0.1,
'gabor_kersize': 16,
'gabor_sig2lam': 0.8,
'gabor_theta': 45.0,
'gabor_thold': 0.025,
'ibsi': 0,
'n_loader_threads': 1,
'n_feature_calc_threads': 4,
'neighbor_distance': 5,
'pixels_per_micron': 1.0}
There is also the option to pass arguments to this function to only receive a subset of parameter values. The arguments should be valid parameter names as string, separated by commas. For example,
from nyxus import Nyxus
nyx = Nyxus(["*ALL*"])
intensityDir = "/path/to/images/intensities/"
maskDir = "/path/to/images/labels/"
features = nyx.featurize_directory (intensityDir, maskDir)
print(nyx.get_params('coarse_gray_depth', 'features', 'gabor_f0'))
will print the dictionary
{'coarse_gray_depth': 256,
'features': ['*ALL*'],
'gabor_f0': 0.1}
8. Using Arrow for feature results
Nyxus provides the ability to get the results of the feature calculations in Arrow IPC and Parquet formats. To create an Arrow IPC or Parquet file, use output_type=”arrowipc” or output_type=”parquet” in Nyxus.featurize* calls. Optionally, an output_path argument can be passed to specify the location of the output file.
from nyxus import Nyxus
import numpy as np
intens = np.array([
[[1, 4, 4, 1, 1],
[1, 4, 6, 1, 1],
[4, 1, 6, 4, 1],
[4, 4, 6, 4, 1]],
[[1, 4, 4, 1, 1],
[1, 1, 6, 1, 1],
[1, 1, 3, 1, 1],
[4, 4, 6, 1, 1]],
[[1, 4, 4, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 6, 1, 1],
[1, 1, 6, 1, 1]],
[[1, 4, 4, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 6, 1, 1]],
])
seg = np.array([
[[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1]],
[[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[0, 1, 1, 1, 1],
[1, 1, 1, 1, 1]],
[[1, 1, 1, 0, 0],
[1, 1, 1, 1, 1],
[1, 1, 0, 1, 1],
[1, 1, 1, 1, 1]],
[[1, 1, 1, 0, 0],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1],
[1, 1, 1, 1, 1]]
])
nyx = Nyxus(["*ALL_INTENSITY*"])
arrow_file = nyx.featurize(intens, seg, output_type="arrowipc", output_path="some_path")
print(arrow_file)
The output is:
some_path/NyxusFeatures.arrow
9. Nested Features Examples
The Nested class is the Python API of Nyxus identifies child-parent relations of ROIs in images with a child and parent channel. For example, consider the following intensity and segmentation images of the parent channel,

Fig 1. Parent channel intensity |
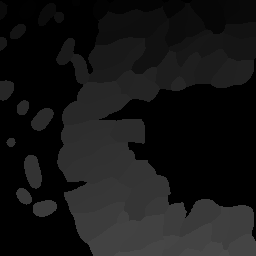
Fig 2. Parent channel segmentation |
With the child channel

Fig 3. Child channel intensity |

Fig 4. Child channel segmentation |
As shown by the figures, there are ROIs in the child segmentation that are completely contained in the the ROIs of the parent channel. The purpose of the Nested class is to identify the child ROIs of the parent channel. The Nested class also contains functionality to apply aggregate functions to the child features, as shown belong in the example.
To use the Nested class, first call the constructor with the optional argument aggregate. If aggregate is not passed, the find_relation behavior will change (described later). Any aggregate function supported by Pandas is available, such as min, max, count, and mean. Lambda functions can also be used, and named using a 2-tuple, where the first element is the name and the second is the lambda function. This allows functions that are not supported by Pandas to be used, such as Numpy’s np.nanmean.
To use the Nested class, first call Nyxus to get the features of all ROIs from the child channels. If the child channels are described by a channel number in the filename, a filepattern can be used to filter down to only the child channel. Consider a directory with the images
p0_y1_r1_c0.ome.tif
p0_y1_r1_c1.ome.tif
p0_y1_r2_c0.ome.tif
p0_y1_r2_1.ome.tif
p0_y1_r3_c0.ome.tif
p0_y1_r3_c1.ome.tif
...
where the child channel is designated by c0 and the parent channel is c1. We can filter down to only the child channel using the filepattern p{r}_y{c}_r{z}_c0.ome.tif or the equivalent regex p[0-9]_y[0-9]_r[0-9]_c0.ome.tif.
Next, we calculate the features for the child channel. For simplicity, we only use the Gabor features, but any or all features can be used.
from nyxus import Nyxus, Nested
import numpy as np
int_path = 'path/to/intensity'
seg_path = 'path/to/segmentation'
nyx = Nyxus(['GABOR'])
child_features = nyx.featurize(int_path, seg_path, file_pattern='p[0-9]_y[0-9]_r[0-9]_c0\.ome\.tif')
print(features.head())
The result of this code is
mask_image intensity_image label GABOR_0 GABOR_1 GABOR_2 GABOR_3 GABOR_4 GABOR_5 GABOR_6
0 p0_y1_r1_c0.ome.tif p0_y1_r1_c0.ome.tif 1 0.224206 0.172619 0.166667 0.730159 0.773810 0.767857 0.753968
1 p0_y1_r1_c0.ome.tif p0_y1_r1_c0.ome.tif 2 1.000000 0.610000 0.540000 0.980000 0.990000 0.990000 0.970000
2 p0_y1_r1_c0.ome.tif p0_y1_r1_c0.ome.tif 3 0.429864 0.217195 0.122172 0.877828 0.941176 0.936652 0.909502
3 p0_y1_r1_c0.ome.tif p0_y1_r1_c0.ome.tif 4 0.846154 0.948718 0.717949 1.000000 1.000000 1.000000 1.000000
4 p0_y1_r1_c0.ome.tif p0_y1_r1_c0.ome.tif 5 0.277778 0.021368 0.029915 0.794872 0.841880 0.841880 0.824786
Next, the find_relation method is used to find the child-parent relations. This method takes in the segmentation path along with filepatterns to distinguish the child channel from the parent channel.
nest = Nested(['sum', 'mean', 'min', ('nanmean', lambda x: np.nanmean(x))])
df = nest.find_relations(seg_path, 'p{r}_y{c}_r{z}_c1.ome.tif', 'p{r}_y{c}_r{z}_c0.ome.tif')
print(df.head())
The result is
Image Parent_Label Child_Label
0 /path/to/image 72.0 65.0
1 /path/to/image 71.0 66.0
2 /path/to/image 70.0 64.0
3 /path/to/image 68.0 61.0
4 /path/to/image 67.0 65.0
The featurize method can then be used along with the child features to apply the aggregate functions. The featurize method takes in the features DataFrame generated by Nyxus, which contains the features calculations for each ROI, along with the DataFrame containing the parent-child relations from the find_relations method. The output of this method is a DataFrame containing
df = nest.featurize(df, features)
print(df.head())
The result is
GABOR_0 GABOR_1 GABOR_2 ... GABOR_4 GABOR_5 GABOR_6
sum mean min nanmean sum mean min nanmean sum mean ... min nanmean sum mean min nanmean sum mean min nanmean
label ...
1 24.010227 0.666951 0.000000 0.666951 19.096262 0.530452 0.001645 0.530452 17.037345 0.473260 ... 0.773810 0.897924 32.060053 0.890557 0.767857 0.890557 31.643434 0.878984 0.753968 0.878984
2 13.374170 0.445806 0.087339 0.445806 7.279187 0.242640 0.075000 0.242640 6.390529 0.213018 ... 0.735000 0.885494 26.414860 0.880495 0.727500 0.880495 25.886468 0.862882 0.700000 0.862882
3 5.941783 0.198059 0.000000 0.198059 3.364149 0.112138 0.000000 0.112138 2.426409 0.080880 ... 0.858462 0.900500 26.836040 0.894535 0.858462 0.894535 26.172914 0.872430 0.829231 0.872430
4 13.428773 0.559532 0.000000 0.559532 12.021938 0.500914 0.008772 0.500914 9.938915 0.414121 ... 0.820175 0.945459 22.572913 0.940538 0.802632 0.940538 22.270382 0.927933 0.787281 0.927933
5 6.535722 0.181548 0.000000 0.181548 1.833463 0.050930 0.000000 0.050930 2.083023 0.057862 ... 0.697917 0.819318 29.094328 0.808176 0.693452 0.808176 28.427727 0.789659 0.675595 0.789659
The other way to utilize the Nested class is to not pass any aggregate features to the constructor. In this case, the featurize method with create a pivot table where the rows are the ROI labels and the columns are grouped by the features.
nest = Nested(['sum', 'mean', 'min', ('nanmean', lambda x: np.nanmean(x))])
df = nest.find_relations(seg_path, 'p{r}_y{c}_r{z}_c1.ome.tif', 'p{r}_y{c}_r{z}_c0.ome.tif')
df = nest.featurize(df, features)
print(df.head())
The result is
GABOR_0 ... GABOR_6
Child_Label 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 ... 55.0 56.0 58.0 59.0 60.0 61.0 62.0 64.0 65.0 66.0
label ...
1 0.666951 NaN NaN NaN NaN NaN NaN NaN NaN NaN ... NaN NaN NaN NaN NaN NaN NaN NaN NaN NaN
2 NaN 0.445806 NaN NaN NaN NaN NaN NaN NaN NaN ... NaN NaN NaN NaN NaN NaN NaN NaN NaN NaN
3 NaN NaN 0.198059 NaN NaN NaN NaN NaN NaN NaN ... NaN NaN NaN NaN NaN NaN NaN NaN NaN NaN
4 NaN NaN NaN 0.559532 NaN NaN NaN NaN NaN NaN ... NaN NaN NaN NaN NaN NaN NaN NaN NaN NaN
5 NaN NaN NaN NaN 0.181548 NaN NaN NaN NaN NaN ... NaN NaN NaN NaN NaN NaN NaN NaN NaN NaN